The biggest challenge for cell and gene therapies
CGTs are safer and more effective than ever - why aren't investors more excited?
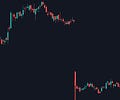
Say I showed you the following two biotech stock charts (same company, each candle represents one trading day). If you had to guess, what type of event would you say each one represents? They both seem fairly negative, particularly the first one, right?
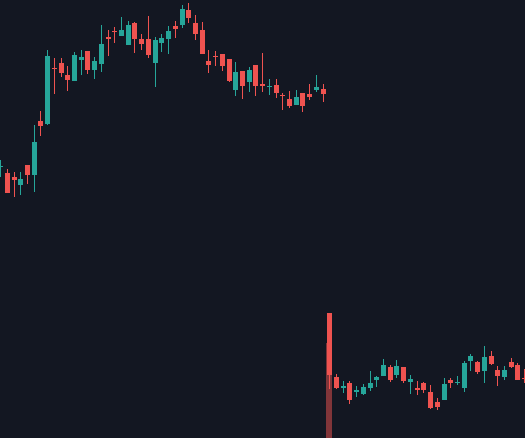
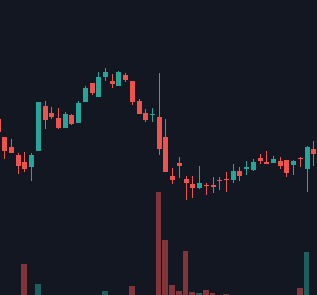
What if I told you that the first chart with the massive gap down represents a FDA Complete Response Letter (CRL - essentially a rejection), while the second one represents a massive FDA approval for the same drug 3 years later? A drug expected to become a rare disease blockbuster mind you. Seems odd doesn’t it?
The charts above are for BioMarin, but a number of recent cell and gene therapy companies have had similar reactions from the market upon approval - mere indifference and skepticism. It’s not like the biopharma industry is doing badly either. Companies with GLP-1 drugs like Eli Lilly and Novo Nordisk are beating the market by wide margins. So what’s really going on?
To answer this we need to take a step back and look at the broader arc of cell and gene therapy development. The field really started in the 60s and 70s and accelerated with the arrival of recombinant DNA techniques, which allowed for the development of cloned genes used to demonstrate that foreign genes could correct genetic defects and disease phenotypes in mammalian cells in vitro.1 By the late 80s/early 90s, scientists were conducting the first gene transfers into humans. In 1999, Jesse Gelsinger received an adenovirus based gene therapy intended to correct a disorder he had been born with: ornithine transcarbamylase deficiency (OTC deficiency), a genetic disease that leads to high levels of ammonia in the blood. He had a mild version of the disease and was able to survive on a special diet and medication. Unfortunately he died 4 days after receiving the gene therapy infusion due to a massive immune response that led to organ failure and brain death.
This event slammed the brakes on the use of gene therapies in humans. Drug developers realized a greater focus on safety and immunogenicity was needed. Although there were gene therapies approved in the following years, it took until 2012 for the EMA to approve the first adeno associated virus (AAV) based therapy Glybera, and until 2017 for the FDA to approve Luxturna (also AAV based). AAV based therapies have become the most popular by far, due to their excellent safety profile (note that AAVs are completely different from adenoviruses despite the similar names). Currently in 2024, the vast majority of gene therapy trials use AAV vectors to deliver the therapeutic material.
Coming back to the original point: the field of gene therapy has advanced significantly since the early days. Gene therapies are much safer than before, and the vector selection/design, therapeutic cassettes, and manufacturing techniques have all improved as well. Gene therapies are potentially transformative because they address the root cause of the disease - the defective protein/enzyme produced due to pathogenic genetic variants.
So why aren’t markets more excited about these innovations?
The short answer is that the markets are skeptical of gene therapy companies’ ability to produce a return on investment. This is true even for companies like Sarepta that have been executing well. Sarepta posted cumulative product sales of $334 million for its Duchenne Muscular Dystrophy (DMD) drug Elevidys in just under a year while its stock has gone nowhere over that time period.
The main challenge companies have been facing is actually not from the drug regulators, but from the payers (insurance companies and public health systems). BioMarin’s hemophilia A drug Roctavian, for example, was expected to fetch at least $1.5-2 million per one-time dose in the EU. However, they reported on an earnings call net revenue of only $900k per dose. In addition to that, despite reporting more than 50 patients potentially eligible for therapy in the EU, only 2 have been dosed there (and that too after a year of delays). In the US only 1 patient has been dosed as of Feb 2024. This has resulted in $3.5 million of revenue vs $50-150 million projected in 2023, a huge miss.2
Clearly, the number of patients, price per dose, and time to dosing are all coming in way under expectations. In addition, it’s unclear exactly how long therapeutic effects will last (durability) as the genetic material being delivered is largely non-integrating into the host genome and therefore may wane in protein expression over time. Payers are having to wrestle with paying for multi-million dollar therapies and many are demanding a ‘warranty’ of sorts, requiring the drug company to pay back or forfeit earnings if the drug does not perform up to a pre-specified level.
So what should drug developers do differently?
Stop thinking “if we build it, they will come” - unmet need and level of competition are critical factors to success.
Gene therapies with a high unmet need, and low competition have so far done best. Therapies like Elevidys for DMD, and Libmeldy for MLD are for conditions which are progressive and fatal and, at the time of launch, had no competing gene therapy or effective standard of care. It should not be surprising that those drugs have hit much higher sales numbers than Roctavian (for severe Hemophilia A) and Hemgenix (for Hemophilia B) despite similar or smaller eligible patient populations.
For companies entering competitive areas, it is all the more critical to sell the story as to why patients should take the risk on a new therapy, and why physicians should prescribe it. Unless it’s the only treatment available for a fatal disease, patients and physicians may be skeptical of switching off of a known and effective standard of care. It is critical to partner with patient advocacy organizations early in the process, especially if the drug company is a new entrant in the field and doesn’t have the trust of the community.
Provide excellent support throughout the entire journey, for all stakeholders
Most aspects of the gene therapy journey are novel and uncertain for all parties involved - the patients, physicians, clinical staff, etc. Unless they participated in the clinical trials, they are probably unfamiliar with all the steps involved in getting the drug, preparing the drug, preparing the patient, administering the drug, etc. Excellent support and education should be provided from start to finish to ensure the process is clear and understood by all key parties.
Start working with payers as early as possible to communicate the value proposition
It’s critical to ensure payers understand the value proposition of the therapy vs alternatives. Recognize that payers are learning on the fly and trying to manage their own risks. Particularly for smaller payers, a single $1-2 million therapy could create a huge burden. Ensure they understand the clinical data and the value of measuring improvements in biomarkers that are directly connected with the disease state. With regard to pricing, it’s important to be strategic and recognize that an agreed upon list price in one region may be used as a benchmark by a payer in a different region.
Think carefully about the global launch strategy
With how fast the field is moving, it shouldn’t be a surprise that health authorities and payers around the world are playing catch up with regulatory frameworks and reimbursement schemes. As demonstrated by Roctavian, winning approval for a gene therapy in the EU did not guarantee commercial success in that region. It’s important to compare the potential benefits from launching a therapy in a region vs the costs and complexity of doing so. For example, the US and Japan have been relatively easier to launch gene therapies in as they are large unified markets whereas the EU has been much more difficult due to varying reimbursement policies between the 27 member states and lower pricing.
The recent commercial struggles with gene therapies are no doubt making companies reevaluate their commercial strategy and choice of therapeutic areas. Introducing a gene therapy into a crowded competitive space with good alternatives makes it an uphill battle. On the other hand, a gene therapy with strong supporting data could make a much bigger impact in a smaller niche where there is less or no competition and a high unmet need.
If you liked this piece, please like/comment/subscribe and share!
https://pubmed.ncbi.nlm.nih.gov/1303270/
https://www.biopharmadive.com/news/biomarin-roctavian-sales-hemophilia-gene-therapy/708321/